Fluid Management
IMPROVING FLUID MANAGEMENT MAY IMPROVE CLINICAL OUTCOMES, POTENTIALLY SAVING MILLIONS IN OPERATING COSTS1-4
IV fluids are among the most commonly used interventions for hospitalized patients, yet 1 in 5 patients on IV fluid therapy suffer complications because fluids were delivered in the wrong volume or at the wrong time.5 The Starling system takes the guesswork out of fluid decisions with dynamic assessments to accurately identify the potential benefits or harms of administering IV fluids to an individual patient.

FLUID RESPONSIVENESS IS DYNAMIC
Every patient has unique and constantly changing fluid management needs. Over 80% of hospitalized patients receive IV fluids.6 Yet studies show that administering too little or too much fluid can lead to serious complications and contribute to rising healthcare costs.7,8
Like IV medications, fluid administration is not a one-size-fits-all approach and should be dosed according to patient response. Assessing whether fluid may help or harm a patient is a critical step in optimizing treatment for the individual. The Starling system can accurately advise clinical staff on whether a patient will benefit from additional fluid, in a 100% non-invasive way.
Starling’s advanced technology meets the dynamic and real-time needs of fluid monitoring.
IV fluids may cause harm.
Fluid is an important predictor of mortality. Only ~50% of hemodynamically unstable patients will respond to IV fluid by increasing cardiac output and perfusion.9
Fluid responsiveness is dynamic.
Fluid status changes over the first 72 hours.10 Using just blood pressure, urine output and heart rate to measure fluid responsiveness may provide delayed and inconclusive information.9,11
Real-time, accurate data is needed.
The Starling fluid management monitoring system provides a full hemodynamic profile within seconds — with 94% sensitivity and 100% specificity for predicting fluid responsiveness in critical care situations.12
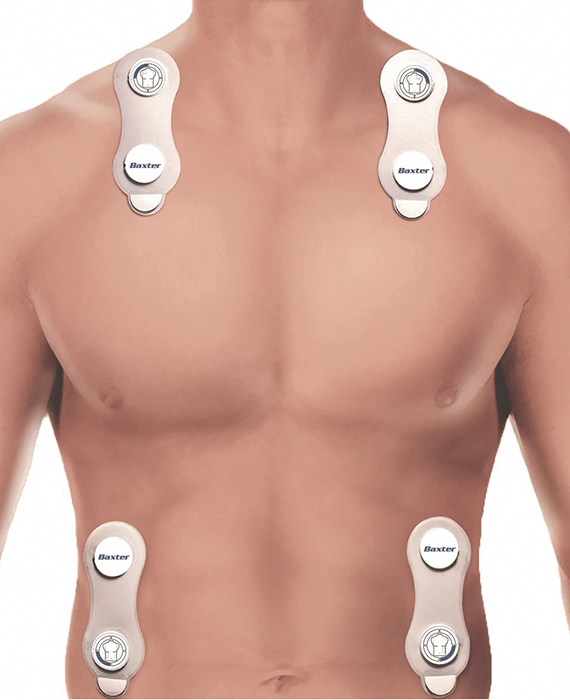
PATENTED AND PROVEN BIOREACTANCE TECHNOLOGY
Starling uses unique, patented Bioreactance™ technology to take measures continuously and precisely, and it requires only four easy-to-place sensor pads. The sensors can be placed anywhere on the chest or back as long as two are positioned above the heart and two are below the heart.
The technology works by sending a small electrical current across the thorax, measuring the time delay or phase shift between the applied electrical current and the voltage on the thorax. These phase shifts correlate with the amount of blood volume change in the thorax; therefore, the stroke volume can be calculated.
